Upon its premiere last year, Zortrax Inkspire was the first resin printer in the company’s history. The device’s UV LCD technology allows the creation of extremely precise, small, and geometrically complex models that can be used in industries, such as architecture, jewelry or medicine. Now Zortrax is taking its next big leap towards the medical market, introducing specialized biocompatible resins that are optimized for the Inkspire 3D printer.
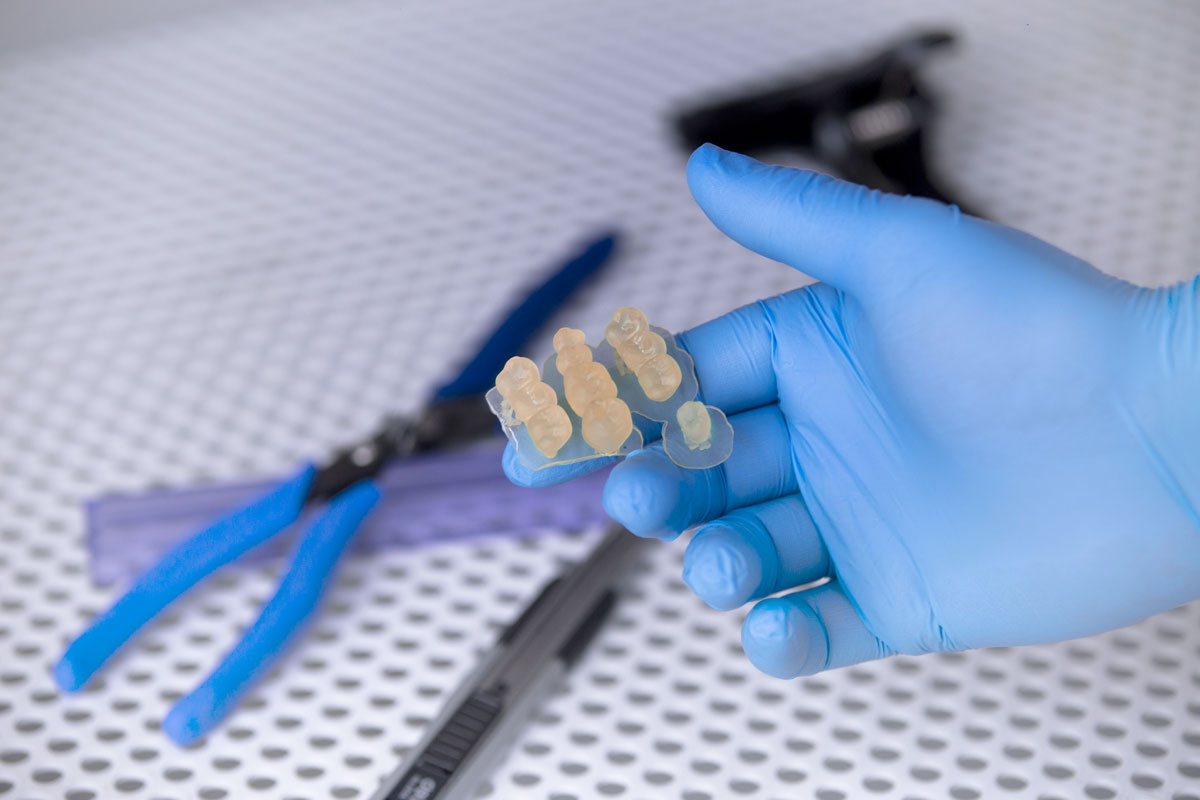
The newly announced resins are Raydent Crown & Bridge, which is used for printing temporary crowns and bridges, and Raydent Surgical Guide for 3D printing surgical guides. The former is class IIa biocompatible, making it safe for contact with human tissue for up to 30 days. It is available
in an A2 shade (beige). High abrasion resistance guarantees permanent smooth surfaces and anatomical shape throughout its use.
Raydent Surgical Guide is intended for 3D printing precise prosthetic surgical guides. Class I biocompatibility makes it safe for transient contact with human tissue, which is enough to carry out procedures commonly performed in modern dentistry. Surgical guides made of this resin are translucent to ensure excellent visibility. High dimensional accuracy guarantees precise placement
of implants or guidance for tools used by a dentist. Low viscosity and water resistance, on the other hand, make the 3D printed guides easy to wash.
Zortrax Inkspire 3D printer has a workspace of 74 x 132 x 175 mm, its XY resolution is 50×50 microns while the minimum layer height is 25 microns. The device prints up to 8 times faster than other resin 3D printers working in comparable technologies (SLA or DLP) and is up to 9 times more precise. Two new biocompatible resins allow Zortrax Inkspire to now be used by prosthetic laboratories not only
in prototyping but also in fabrication of final products meant for intraoral use by patients. This
is a huge step forward for Zortrax, which puts it at the absolute forefront of 3D printing solutions for dentistry and prosthetics.
Subscribe to our Newsletter
3DPResso is a weekly newsletter that links to the most exciting global stories from the 3D printing and additive manufacturing industry.