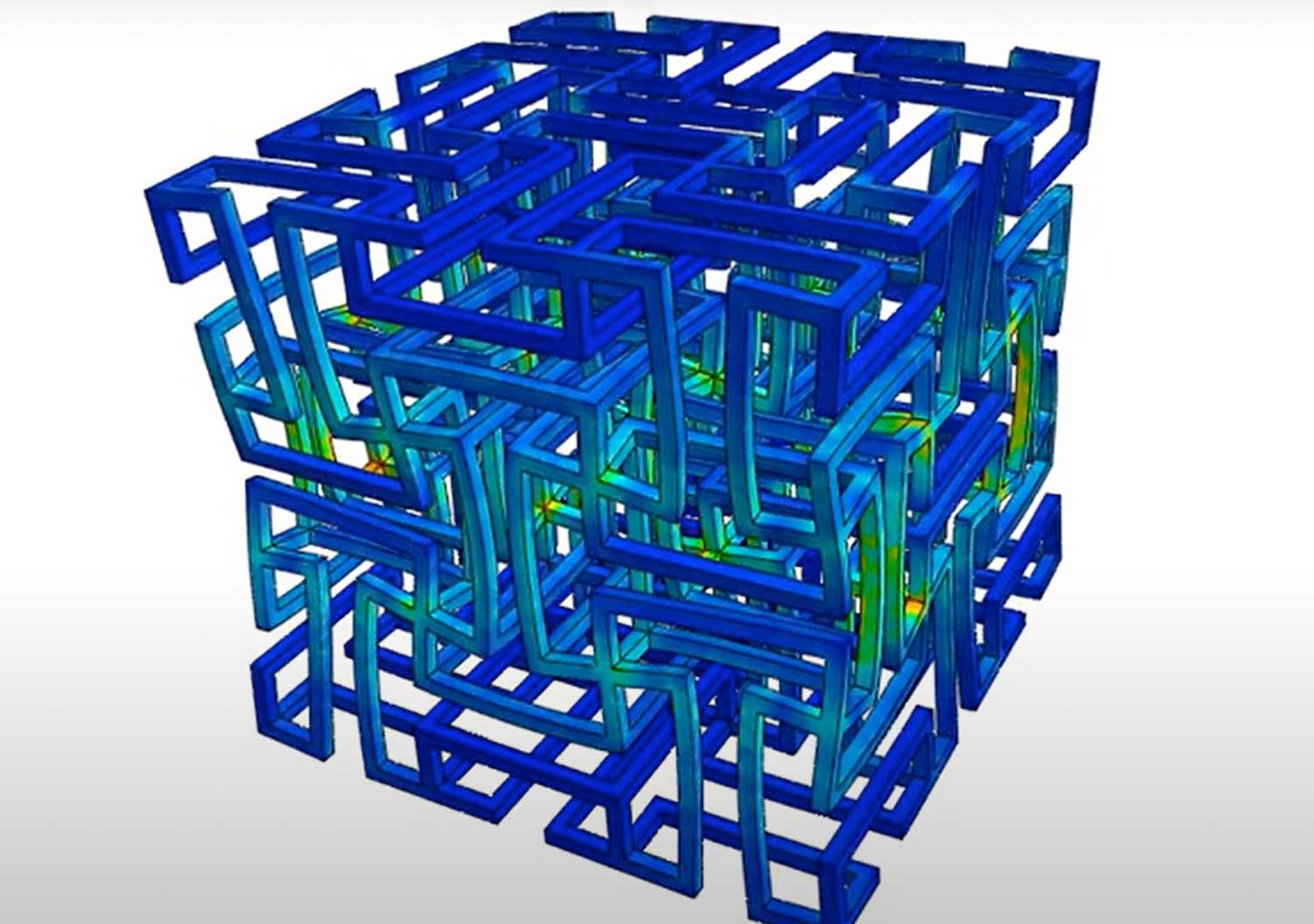
Researchers are using architectural tools to achieve 300 times more flexibility in a new 3D printed design.
In the lab of Georgia Tech researcher Scott Hollister, 3D printed medical devices are being created that have already saved the lives of many young patients. For over a decade, Hollister and his team have been developing life-saving, patient-specific airway splints for babies with rare birth defects.
These customized airway support devices are made from a biocompatible polyester called polycaprolactone (PCL), which is approved by the U.S. Food and Drug Administration. The manufacturing process uses selective laser sintering to heat the polyester powder, which then bonds into a solid structure. PCL-based devices have demonstrated a high level of safety when implanted in patients.
However, PCL has the disadvantage of having relatively stiff and linear mechanical properties, which has limited its application in other important areas of biomedicine, such as soft tissue engineering. However, Hollister’s lab has figured out how to turn the rigid thermoplastic into a flexible material that can potentially grow with the patient.
Jeong Hun Park, a researcher in Hollister’s lab, conducted a study that demonstrated the successful 3D printing of PCL for soft tissue engineering. An auxetic material, unlike typical elastic materials, has a negative Poisson’s ratio. This means that when an auxetic material is stretched longitudinally, it also expands laterally, whereas most materials constrict laterally (as they have a positive Poisson’s ratio).
“Although the mechanical properties and behavior of the 3D structure depend on the inherent properties of the base material — in this case, PCL — it can also be significantly tuned through internal architecture design,” explained Park.
The research of Hollister’s team aims to give PCL new auxetic properties. Park explains that the mechanical properties and behavior of the 3D structure not only depend on the inherent properties of the base material – in this case PCL – but can also be significantly influenced by the design of the internal architecture.
The team began by creating cube-shaped structures to test the flexibility, strength and permeability of the auxetic design. An auxetic material is a network structure built by assembling single cells. These single cells consist of struts and their intersecting joints, which are an important feature of the behavior of an auxetic device.
The rotation of these cross-joints in the network under compression or strain results in negative Poisson’s ratio behavior. This enables advanced performance for a printed device, including impact energy absorption, resistance to indentation and high flexibility.
“When you look at the numbers, based on Jeong Hun’s work, the new structure is about 300 times more flexible than the typical solid structure we make out of PCL in our lab,” said Hollister, professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University, where he also holds the Patsy and Alan Dorris Chair in Pediatric Technology and serves as the department’s associate chair for translational research.
The ultimate goal of the research is to use this structure to develop a breast reconstruction implant that has comparable biomechanical properties to native breast tissue. These biodegradable breast reconstruction implants serve as a kind of scaffold, whereby the biocompatible material (PCL) is eventually degraded and absorbed by the body while retaining similar mechanical properties to native breast tissue.
The 3D printed breast implant is designed to provide reconstructive support while promoting the growth of new tissue. The space between the tiny struts makes all the difference to the larger device, allowing for softness and pliability that would otherwise have been impossible. These spaces can eventually be filled with hydrogel, which promotes the growth of cells and tissue.
The architecture designed by Park also includes the design of internal voids and spaces within the struts, creating a type of microporosity that allows for the mass transport of oxygen, nutrients and metabolites to promote the expansion and growth of a cellular network.
“We expect that native tissue will be first infiltrated into the pores of the biodegradable implant,” Park said. “Tissue volume will then increase within the implant as it degrades and eventually the device itself is replaced with the tissue after complete degradation of the implant.”
Park is working with Emory surgeon Angela Cheng to submit a grant proposal for further research and testing of the breast implant. The team is already adapting the technology for other applications. For example, Mike Davis’ lab at Emory University, which focuses on regenerating heart muscle tissue, is using the technology to reconstruct infarcted or necrotic myocardial tissue.
“The advantage there is, with this design, it can expand in two directions,” he said. “So, as young patients grow, the new device will grow with them.”
Subscribe to our Newsletter
3DPresso is a weekly newsletter that links to the most exciting global stories from the 3D printing and additive manufacturing industry.