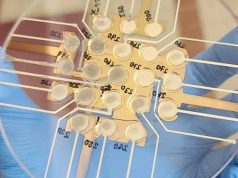
3D bioprinting company Organovo has just reported its financial results for the fiscal year ended March 31, 2015 as well as its total contract bookings for the exVive3D human liver tissue.
After the announcement the commercial release of exVive3D for preclinical drug testing back in November 2014, clients have been able to to access the technology through Organovo’s contract research services program. So far the company has recorded total contract bookings of approximately $ 1.94 million. Customers include multiple top 25 global pharma companies, additional public pharmaceutical companies from small to large cap and private venture-backed companies. Contracts have been signed with customers in the US, Europe and Asia.
Keith Murphy, CEO of Organovo commented: “Response to the exVive3D Human Liver Tissue has been strong, and in line with our expectations. We continue to expect this tissue to grow into the tens of millions in annual revenue, and that it has $100M+ revenue potential. The fact that recent data shows that the exVive3D Human Liver Tissue clearly demonstrated the toxicity of another drug missed during preclinical studies, troglitazone (Rezulin), which exhibited a clear response by causing a statistically significant drop in liver function in our liver tissue at 7 days, provides the potential for our commercial penetration to accelerate even faster.”
Its exVive3D human liver tissue is not the only product the company is currently focussing on. At the 2015 Experimental Biology Conference in April, Organovo released first results of their 3D printed in vitro kidney tissue, using the company’s proprietary NovoGen bioprinting platform.
Organovo has also entered exclusive research collaborations with the pharmaceutical company Merck as well as with L’Oreal USA. According to Murphy, the company is expecting additional partnerships of this kind in the future.
Subscribe to our Newsletter
3DPresso is a weekly newsletter that links to the most exciting global stories from the 3D printing and additive manufacturing industry.