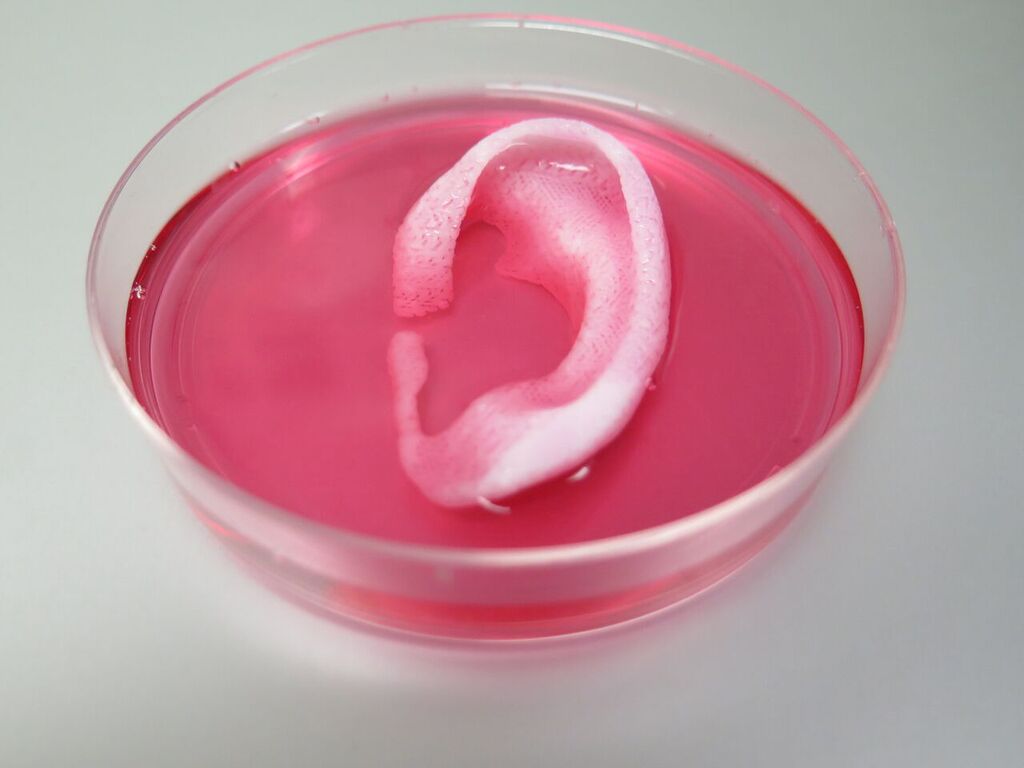
A team of scientists from Wake Forest Baptist Medical Center has developed a 3D bioprinter to produce human-scale tissue constructs such as cartilage, bones and muscle. The study was published in the Nature Biotechnology journal.
The 3D printed constructs were created using their Integrated Tissue-Organ Printer (ITOP) and have already been implanted into animals. The results were promising, showing that the structures have the strength, size and function for use in humans. So far, the major challenge was to produce structures that are stable enough for implantation.
During the process hydrogels containing cells and biodegradable polymers are printed in patterns based on imaging data, before being coated with a temporary polymer as an outer shell. This results in a stable, implantable structure that could potentially be used for clinical applications. While the polymer materials will slowly degrade, the supporting structure helps to maintain the shape until the cells have reorganised themselves.
“This novel tissue and organ printer is an important advance in our quest to make replacement tissue for patients,” said Anthony Atala, director of the Wake Forest Institute for Regenerative Medicine (WFIRM) and senior author on the study. “It can fabricate stable, human-scale tissue of any shape. With further development, this technology could potentially be used to print living tissue and organ structures for surgical implantation.”
So far, the 3D printed ears, muscle and bone tissue have been implanted under the skin of mice and rats, showing promising results. The ears kept their shape and cartilage tissue as well as blood vessels were formed after two months. The implanted printed muscle tissue was robust enough to maintain its structure, has become vascularised and induced nerve formation after two weeks. The human-sized jaw bone fragments printed from human stem cells has formed vascularised bone tissue after five months.
“Our results indicate that the bio-ink combination we used, combined with the micro-channels, provides the right environment to keep the cells alive and to support cell and tissue growth,” said Atala. Currently ongoing studies will focus on the long-term results.
Subscribe to our Newsletter
3DPresso is a weekly newsletter that links to the most exciting global stories from the 3D printing and additive manufacturing industry.