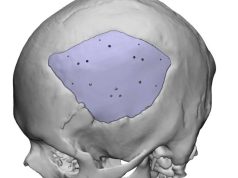
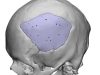
ChoiceSpine, a company specializing in medical applications for the spine, recently received approval from the U.S. Food and Drug Administration (FDA) for the Blackhawk Ti 3D-printed Cervical Spacer System. This system, which utilizes BioBond 3D-printed titanium technology, provides a solution for patients requiring anterior cervical treatment.
It features pre-assembled integrated anchor technology and a proven cam-locking mechanism, and is available in two anatomical footprints that provide significant space for graft material.
ChoiceSpine is known for excellent products and positive customer experiences. The addition of the Blackhawk Ti system is well received by industry experts.
“The Blackhawk Ti Standalone Cervical Spacer System provides excellent intra and post-operative results for my patients requiring the anterior cervical approach,” said Dr. Anders Cohen, DO, Lead Design Surgeon. “The wide variety of implant footprints and profiles allows for restoration of disc height and optimal sagittal balancing. Furthermore, what sets the Blackhawk® Ti apart from others is the simplicity of the implant design with its integrated bone anchors and instrumentation that help reduce surgery time by decreasing the number of steps in the procedure.”
“The expanded indication of the Blackhawk Ti Standalone Cervical Spacer System is the natural progression for the product platform,” states Steve Ainsworth, PhD, ChoiceSpine’s Executive Vice President of Strategy and Technology. “Blackhawk Ti is the first to market a 3D printed system that utilizes preassembled integrated anchor technology. Blackhawk Ti is just one of ChoiceSpine’s numerous 3D printed titanium fusion solutions. This standalone clearance is an excellent example of how ChoiceSpine brings technically superior spinal devices to the market and does Spine the Right Way℠.”
The Blackhawk Ti system incorporates ChoiceSpine’s proprietary 3D-printed BioBond technology and offers wide margins for bone material and maximum visualization. ChoiceSpine relies on a collaborative team of physicians and industry partners to bring technically superior products to market.
Altus Capital Partners is an investment firm focused on mid-market manufacturing companies and has a unique perspective on the opportunities and challenges of this industry due to its exclusive focus on the manufacturing space.
This FDA approval is a significant step and underscores the progress being made in the development of spinal devices manufactured using 3D printing technologies.
Subscribe to our Newsletter
3DPResso is a weekly newsletter that links to the most exciting global stories from the 3D printing and additive manufacturing industry.