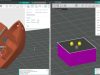
Sartorius uses 3D printing to produce disposable components for the biopharmaceutical industry. According to Sartorius, the additive process is ideal for low-cost, small-batch production of biocompatible parts.
As a supplier to vaccine and drug manufacturers, Sartorius is subject to strict regulatory requirements. For use in sensitive production, all components must meet the highest standards in terms of biocompatibility and traceability.
According to Sartorius, the certified production at the service provider Materialise meets these conditions. So far, more than 26,000 disposable components made of PA12 plastic have been printed for customers in this way. In addition to conformity to standards, Sartorius also appreciates Materialise’s responsiveness in adapting the process.
For the highly regulated medical technology industry, Sartorius sees great potential in 3D printing if the technology is used sensibly and moderately. The goal is to save time and costs through additive manufacturing and to enable new geometries.
Subscribe to our Newsletter
3DPresso is a weekly newsletter that links to the most exciting global stories from the 3D printing and additive manufacturing industry.